Probing a Parkinson’s Paradox


In one video, an elderly gentleman who normally can barely walk pedals a bike effortlessly down an Amsterdam street. In another video, a man who typically can’t hold a mug without sloshing the water out of it catches a ball as he jogs across the room and tosses it back to the person who threw it. This phenomenon of swift, smooth movement in patients whose motor functions are impaired by Parkinson’s disease remains a mystery to the scientists who study the disorder and the part of the brain it affects.
“It’s called paradoxical kinesia, which means paradoxical movement,” said Scott Grafton, a UC Santa Barbara neuroscientist who studies how people organize movement into goal-oriented action. “People with Parkinson’s can be frozen most of the time, and then every once in a while whatever brakes are on in their motor system are released and they move normally.”
The underlying problem with Parkinson’s disease is that the circuit in the brain that governs movement is disrupted, Grafton explained. So where does the ability to ride a bike or catch and throw a ball while jogging come from?
Thanks to the Aligning Science Across Parkinson’s (ASAP) initiative, Grafton and Regina Lapate, an assistant professor in the Department of Psychological & Brain Sciences, will get us closer to finding out. The $12 million, three-year, multi-institution project led by the University of Pittsburgh allows scientists like Grafton, Lapate and their colleagues at Pitt to explore and investigate brain circuits that are involved in Parkinson’s disease and little-understood phenomena such as the placebo effect, and paradoxical kinesia, which have patients moving despite the degeneration of their motor circuits.
A Deep Mystery
The presence of coordinated movement in Parkinson’s patients, however limited, suggests that there is some integrity left in the basal ganglia, Grafton said. These structures are found deep in the brain and are associated with the initiation and execution of movements, orchestrating the multitude of signals that tell some muscles to move while inhibiting competing actions. In patients with Parkinson’s, these nerve clusters are destroyed and stop producing the neurotransmitter dopamine. The result is a variety of physical symptoms, including tremors while standing still, poor balance and slowness or the cessation of movement.
“You get a lot of noise and bad rhythms in the motor circuit,” Grafton explained. In treatments involving deep brain stimulation, pulses of electricity to a node in this network are able to clear the jam and restore some voluntary motor function in patients and improve responsiveness to dopamine replacement medicines.
Meanwhile, the placebo effect, reported often in clinical and surgical trials, is just as it suggests: Some patients who believe they had been treated by medication or a procedure are able to move, despite not actually having been treated.
It’s still unclear where the coordinated movement comes from, but the scientists have some ideas.
“The way we’re thinking about is there’s this secret door or alternative channel that comes and influences all of this network,” Grafton said. “And that’s the one that is essential for both the placebo effect and paradoxical kinesia.”
The scientists’ efforts focus on two neuronal circuits: a closed loop that travels from the basal ganglia deep in the brain to the motor cortex in the frontal lobe and back; and an open loop that receives input from the amygdala — the circuit researchers suspect could hold the secret door.
The amygdala in particular captures the attention of Grafton and Lapate, who will focus on identifying and investigating the open loop circuit.
“The amygdala is often described as an early warning system,” Grafton explained, adding that it detects threat and activates appropriate behaviors in response to dangerous stimuli, sometimes even before the person knows it.
“If you’re running down a trail, and there’s a wiggly, squiggly thing on it, it’s going to make you jump over it before you even know it’s a snake,” he said. “So, it’s a great system for the quick release of specific movements that are appropriate to whatever the threat is.”
Part of this threat perception performed by the amygdala is the experience of heightened emotions such as fear, stress and anxiety — responses that fall within Lapate’s area of expertise.
“In the intriguing phenomenology of paradoxical kinesia, Parkinson’s patients’ movement, normally impaired, is facilitated by surprising or emotionally evocative situations that require a fast response — for example, rescuing a child who is falling off a high chair,” Lapate explained.
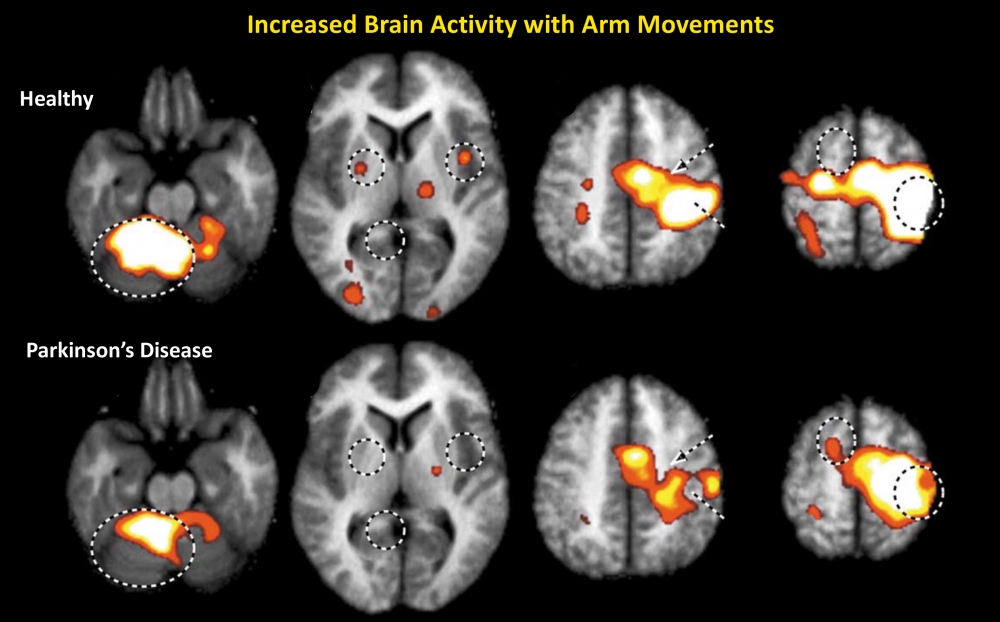
To understand this potential amygdala-dependent motor circuitry, she and Grafton have designed a series of tasks that employ emotionally evocative stimuli, and that require motor output at specific time windows while acquiring continuous measurements of the activity within the whole brain via functional MRI.
“This will allow us to probe whether amygdala function — its increased engagement, heart rate mobilization in emotionally evocative situations and its hypothesized emotion-dependent connectivity with alternative motor structures believed to be part of this circuitry — explains emotion-dependent gains in motor function,” Lapate said.
The UCSB scientists’ work complements the neurobiological studies conducted by their colleagues at University of Pittsburgh: mapping both neuronal circuits (Peter L. Strick); manipulating neuron activity in the two circuits in normal and parkinsonian monkeys (Robert Turner); examining dynamic aspects of dopamine release in the monkeys (Helen Schwerdt); and characterizing cell-type-specific gene expression and regulation in the two circuits before and after dopamine depletion (William R. Stauffer).
The information gathered from these studies will contribute to an ever-growing body of knowledge about our brains and Parkinson’s disease, and could also lead to better and new therapies for the millions of people worldwide who live with the disorder.
“We hope that by shedding new light into the function of this little-understood alternative motor circuitry in humans, our work may point to novel future targets for treatment to ameliorate motor dysfunction in Parkinson’s disease,” Lapate said.
Aligning Science Across Parkinson’s is a coordinated research initiative to advance targeted basic research for Parkinson’s disease. Its mission is to accelerate the pace of discovery and inform the path to a cure through collaboration, research-enabling and data sharing. The Michael J. Fox Foundation for Parkinson’s research is ASAP’s implementation partner and issued the grant.