
Clot Buster

In a potential paradigm change for sepsis diagnostics, a new test predicted sepsis soon after infection in mice — well before blood clotting and organ failure — enabling early antibiotic treatment and markedly increased survival. The findings provide a platform to develop rapid and easy-to-perform clinical tests for early sepsis detection and clinical intervention in human patients.
The collaborative effort by a research team including scientists from UC Santa Barbara, UC San Diego, and Sanford Burnham Prebys Medical Discovery Institute (SBPMDI) is detailed in a new study published in the Lancet journal, eBioMedicine. The team succeeded in detecting a catastrophic shift in blood protein abundance soon after infection that can predict sepsis well before disease symptoms and organ damage arise.
The project was led by professor Michael Mahan of UC Santa Barbara, along with professors Dzung Le of UC San Diego, and Jeffrey Smith and Jamey Marth of SBPMDI. Additional collaborators include UCSB scientists Douglas Heithoff and Scott Mahan, as well as SBPMDI scientists Genaro Pimienta and Won Ho Yang, and University of Sydney veterinarian scientist John House.
Sepsis is the number one cause of death in U.S. hospitals. In the clinic, sepsis is diagnosed by a symptom-based approach that may include kidney or liver failure, blood clotting or bleeding — which often occurs well after permanent organ damage. Thus, molecular diagnostics that detect infection at early stages of disease to minimize host injury are sorely needed.
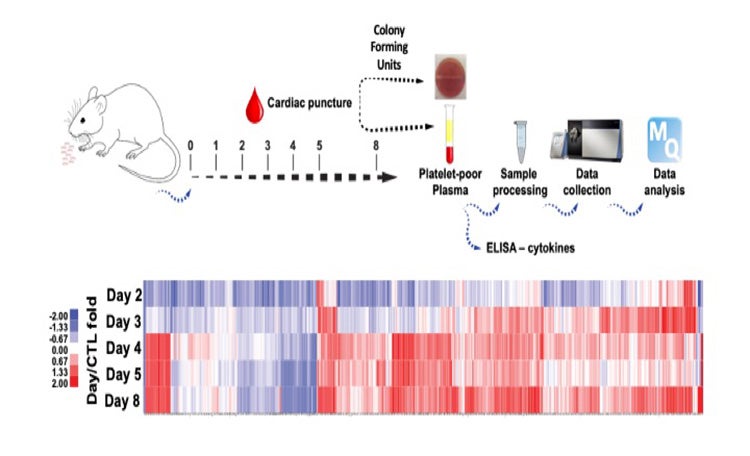
Bacterial infection results in catastrophic changes in blood proteins that can be used as a platform to develop rapid and easy-to-perform tests to predict sepsis for early intervention
Photo Credit: COURTESY IMAGE
“The key finding was identifying proteins in the blood that arise very soon after infection — well before overt disease symptoms,” Mahan explained. “Early detection is critical for clinical intervention to increase survival in sepsis patients.”
To carry out the test, a small amount of blood was collected and analyzed for an increase in coagulation proteins that are induced but inactive at early stages of infection. Such detection enabled early antibiotic treatment — well before activated coagulation proteins induced blood clotting — resulting in markedly increased survival in mice. The technology is open source and freely accessible to all.
The study also demonstrated that antibiotics are less effective after blood proteins increase in response to infection. Treatment failure may be due to host injury triggered by excessive blood clotting, providing insight into why delays in antibiotic treatment in human sepsis are associated with increased risk for death.
“The future plan is to identify a biopanel of early sepsis blood proteins for incorporation into existing blood tests, enabling sepsis prediction well before excessive blood clotting and permanent organ damage,” Heithoff explained.
The researchers demonstrated that the changes in blood proteins soon after infection observed in mice were similar to that reported for human sepsis. Thus, they are optimistic that these findings are translatable for the early detection and treatment of sepsis in humans.
“Currently, one in four patients die of sepsis, with many survivors experiencing lifelong debilitation with cognitive decline,” Scott Mahan said. “We hope technologies like this offer new ways of delivering state-of-the-art molecular diagnostics that predict sepsis before permanent injury occurs.”
This research was funded by grants from the National Institutes of Health’s National Heart, Lung, and Blood Institute, Santa Barbara Cottage Hospital, and the U.S. Army Research Office via the Institute for Collaborative Biotechnologies cooperative agreement and contract.
Study title: “Coagulation Factor Protein Abundance in the Pre-septic State Predicts Coagulopathic Activities that Arise During Late-stage Murine Sepsis”




