Filtering Out the Noise

It’s a familiar situation: You’re talking to a friend at a restaurant, and despite the background din, you still hear each other clearly. Obviously, our brains are capable of filtering out noise, but scientists are still learning how.
Using fruit flies as a model, Luis Franco, a postdoctoral fellow at UC Santa Barbara, set out to investigate the mechanisms underlying this phenomenon. He and his colleague, Emre Yaski at the Kavli Institute for Systems Neuroscience, in Norway, discovered that the background activity in a fly’s brain quieted down when exposed to a familiar odor. This response enhanced the signals specific to the odor of interest. Their results, which appear in the journal Cell Reports, provide a more complete account of what happens when an animal’s brain focuses on processing a particular sensory cue.
Our brains are constantly processing input from our senses: arranging it, filtering it and synthesizing it to provide us with an account of our environment. “However, we didn’t have many details about how this happens in the brain mechanistically,” Franco said.
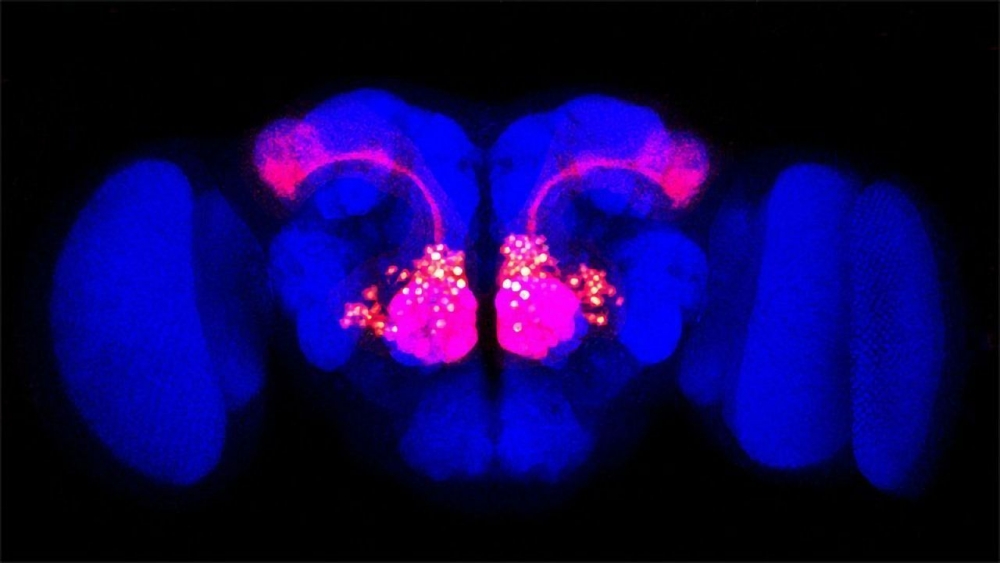
The researchers worked with genetically modified fruit flies that could give them a unique view of neural activity. Neurons in the animals’ antennal lobe — which processes olfactory information — contained a gene that codes for a fluorescent protein. When a neuron is activated, the fluorescence increases; when a neuron is depressed, the fluorescence decreases, Franco explained.
The scientists repeatedly exposed flies to one of eight different odors. These ranged from 1-pentanol (an apricot-like smell) to 2- heptanone (a blue cheese-like smell). Then, after six to 10 minutes, they reintroduced the animals to those same odors.
When a fly wasn’t focused on anything, its background neural activity made the whole lobe twinkle. But when the fly latched onto a particular odor, ongoing activity quieted down, and only neurons with an affinity for the odor lit up. Before this study, scientists hadn’t known how this was happening or how this process enhanced sensory representations. To Franco, the results suggest that an animal’s ability to focus on sensory input relies as much on overall activity decreasing as it does on activity in specific pathways increasing.
As a next step, Franco and Yaski repeated the experiment, but this time they introduced a second odor a single time. When they tested the fly later, they found that the more familiar odor elicited responses with a greater signal-to-noise ratio in the fly’s brain. The difference suggested that the animal was better able to discriminate it from the other smell.
Fruit flies are particularly smell-driven, which is why Franco and Yaksi looked at activity in the olfactory region of the animals’ brains. “But the mechanism we found in flies also applies to neural circuits in other animals, including humans,” he said.
Franco suspects a comparable process occurs in most situations where an animal needs to focus on specific sensory information. “There is a similar process involved with visual information,” he said. “For instance, when you’re driving and focusing on the road ahead, filtering out the noise of other sensory cues is important to properly process the most relevant current information.”
The next step in this line of research would be to identify the specific neurons that mediate this response. Some of Franco’s colleagues have also developed a method for conducting similar experiments with unrestrained flies over the course of multiple days. These techniques could help scientists investigate how this neurological filter affects memory and behavior.
Franco now works with mice, which he said are a more suitable model for many of the questions he wants to ask. “Ultimately I want to understand how the brain integrates information from the environment, compares it to internal reference frames and produces appropriate behavioral outputs.”



