
Unusual Building Blocks

The animal kingdom abounds with creatures that grow hard shells, carapaces and skeletons. But complex life was pretty squishy when it first evolved, so when and how did this change?
UC Santa Barbara paleontologist Susannah Porter and her colleagues tackled this question and found that when carbonate skeletons were first evolving more than 500 million years ago, diverse groups of animals all converged on a similar, counterintuitive process for biomineralization.
The researchers’ findings appear in a new paper published in the Proceedings of the National Academy of Sciences.
Today many unrelated animals build their skeletons or shells out of calcium carbonate — including echinoderms, mollusks and corals. Instead of building crystals ion-by-ion from the surrounding sea water, these animals use amorphous, or non-crystalline, nanoparticles as their building blocks of choice.
“In fact, crystallization by particle attachment actually seems to be the prevailing method of biomineralization as far as we can tell,” said Porter, a professor of earth science.
Rather than building their skeletons at a molecular level, these animals first form nanoparticles of amorphous calcium carbonate. They then store these particles in vesicles that can be used to transport them to the site of crystallization.
This method of crystallization was first documented more than 20 years ago in the teeth of sea urchins. Since then, scientists have noticed the process throughout the animal kingdom, and involving different minerals. What’s more, the different groups of animals seem to have independently settled on this method of biomineralization, so it must have something going for it.
Given its ubiquity, Porter and her collaborators wanted to determine how far back they could find evidence of this process. “We obviously can’t watch these Cambrian and Ediacaran organisms make their skeletons, so we need to have a proxy,” she said. Porter’s colleague, first author Pupa Gilbert, of the University of Wisconsin-Madison, had previously found that crystallization by particle attachment leaves an irregular particulate texture in the shells and skeletons when they’re viewed under a scanning electron microscope.
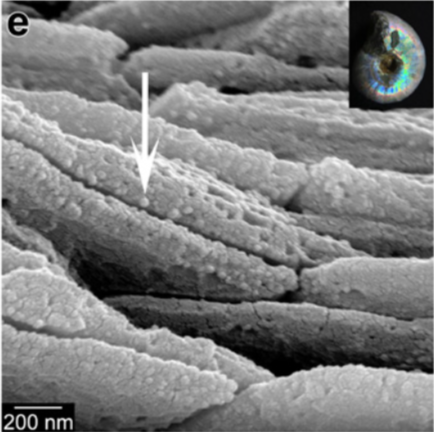
A cretaceous ammonite displays the grainy texture of crystallization by particle attachment when viewed under a scanning electron microscope.
Photo Credit: SUSANNAH PORTER
The team saw this same telltale pattern upon imaging fossils more than 500 million years old. In fact, this signature was preserved even in material that had subsequently converted into another mineral.
“It’s spectacular,” Porter exclaimed, “the fact that we can see this detail at the sub-micrometer level.”
Among the ancient material Porter and her collaborators examined were fossils of Cloudina, a genus that includes some of the earliest animals that formed a mineralized skeleton. The genus was named after UC Santa Barbara’s own Preston Cloud, the late professor of biogeology, and preeminent researcher in the study of early life.
The team saw the same irregular nanoparticulate texture in Cloudina fossils as in other animals that form crystals by particle attachment. “This shows that, even when animals were first evolving mineralized skeletons, and were maybe not so good at biomineralizing, they were already choosing this mechanism,” Porter said.
The researchers’ findings suggest that, even early on, there was selection for this particular mechanism across different lineages. “When you see something that is selected for over and over again, it suggests that it is the most advantageous one,” Porter said.
Although it’s counterintuitive that animals would use amorphous material to create the crystals that ultimately form their skeletons or shells, Porter explained that this mechanism seems to permit greater control over mineralization than simply building ion by ion, as the traditional models suggested.
For one, these particles are incredibly stable when confined in vesicles: The material doesn’t immediately crystallize but remains amorphous. This allows the animal to keep ingredients around and available yet maintain flexibility regarding when and where the mineralized skeleton forms.
Additionally, compounds like calcium carbonate can take different structures — thereby forming different minerals — depending on environmental conditions. By storing the molecules in an amorphous state, the animal can better control what form, or polymorph, they become, Porter explained.
“It’s like having some frozen cookie dough around that you’re later going to bake into cookies,” she said.
Porter is interested in the large-scale patterns of when lineages first evolved skeletons and how those skeletons were affected by the environmental and ecological conditions of the time. She recently submitted a paper looking at how the patterns of carbonate biomineralizers shifted between the Ediacaran and Cambrian periods, when complex animals began to appear in the fossil record.
She suspects that the earliest biomineralizers, like Cloudina, didn’t have particularly strong control over the process of building their skeletons. “But by the time you get to the Cambrian, the carbonate mineralizers have shells that are complex and organized,” said Porter. “They have much greater control over their skeletons.”



